Software To Help You Turn Your Data Into AI
Forget fragmented workflows, annotation tools, and Notebooks for building AI applications. Encord Data Engine accelerates every step of taking your model into production.
Contents

In the scientific, and especially data science community, the word “experiment” means to test a hypothesis until empirical data agrees or conflicts with an experiment's desired outcomes. Machine learning medical imaging experiments need to be rigorous.
In medical imaging machine learning experiments, this involves testing dozens of datasets using machine learning models to achieve higher levels of accuracy, until the artificial intelligence model can be put into production.
Running medical imaging dataset experiment is an essential part of building a stable, robust, and reliable computer vision model (such as a tool for use in oncology). The outcomes of these experiments are even more important when building models for healthcare; you have to be even more confident in the accuracy of the results, as this could influence a life-or-death decision for patients.
However, running multiple experiments can quickly become a massive challenge. Managing the models, datasets, annotators, and experiment results is a full-time job. An inefficient workflow for managing these experiments can make these problems much worse.
In this article, we will look at how to increase the efficiency and effectiveness of your medical imaging dataset experiments to create state-of-the-art models.
Running experiments for machine learning and computer vision models is crucial to the process of creating a viable and accurate production model. At the experimental stage, you need to figure out which approach will work and which won’t.
Once you’ve got a working model and a source of ground truth (dataset), then you can scale and replicate this approach during the production stage to achieve the project outcomes and objectives.
Reaching this goal means going through dozens of experiments. It’s a time-consuming task, and running experiments is a full-time job. You need a team of annotators, a large volume of high-quality data (medical imaging datasets of tumors or lesions, for example), and the right tools to make this work easier. At every stage, the results should gradually improve, until you’ve got a viable and accurate model and process.
Before starting any experiment cycle, it’s important to know the key parameters you want to track. For example, hyperparameters, model architectures, accuracy scores, loss measures, weighting, bias, gradients, dependencies, and other model metrics. Once the experiment outcomes, goals, and metrics are clear, then you can start running machine learning imaging dataset experiments.
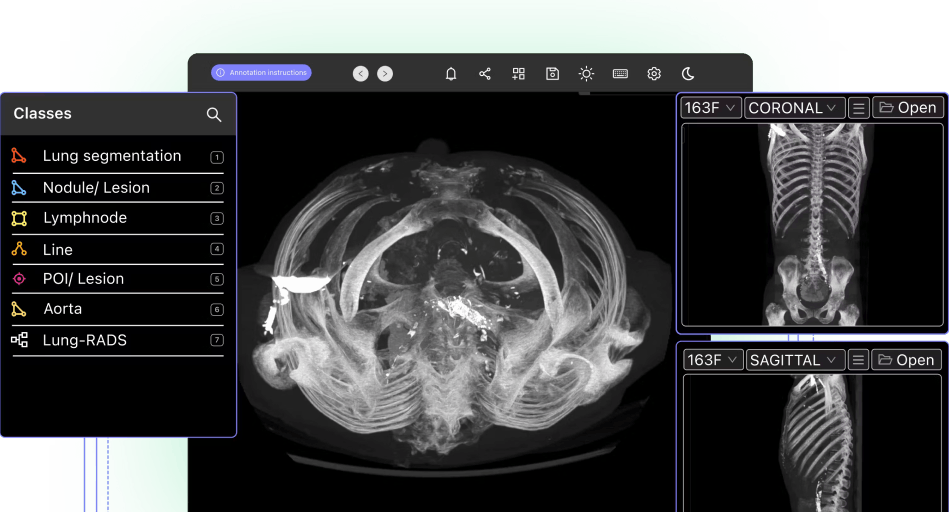
In the healthcare sector, medical image machine learning and computer vision models play an integral role in patient diagnosis, our understanding of diseases, and numerous other medical fields.
Medical images come from numerous sources (including Magnetic Resonance Imaging (MRI), X-rays, and Computed Tomography (CT images)) for a range of conditions, such as Alzheimer's disease, lung cancer, or breast cancer. Unlike other datasets in other sectors, medical images come in more complex formats, including DICOM and NIfTI. These widely-used medical image file formats have several layers of data, such as patient information, connections to other databases, and appointment details.
Even when patient training data is anonymized, the layers and formats make medical imaging datasets more detailed and involved than you will find in other sectors.
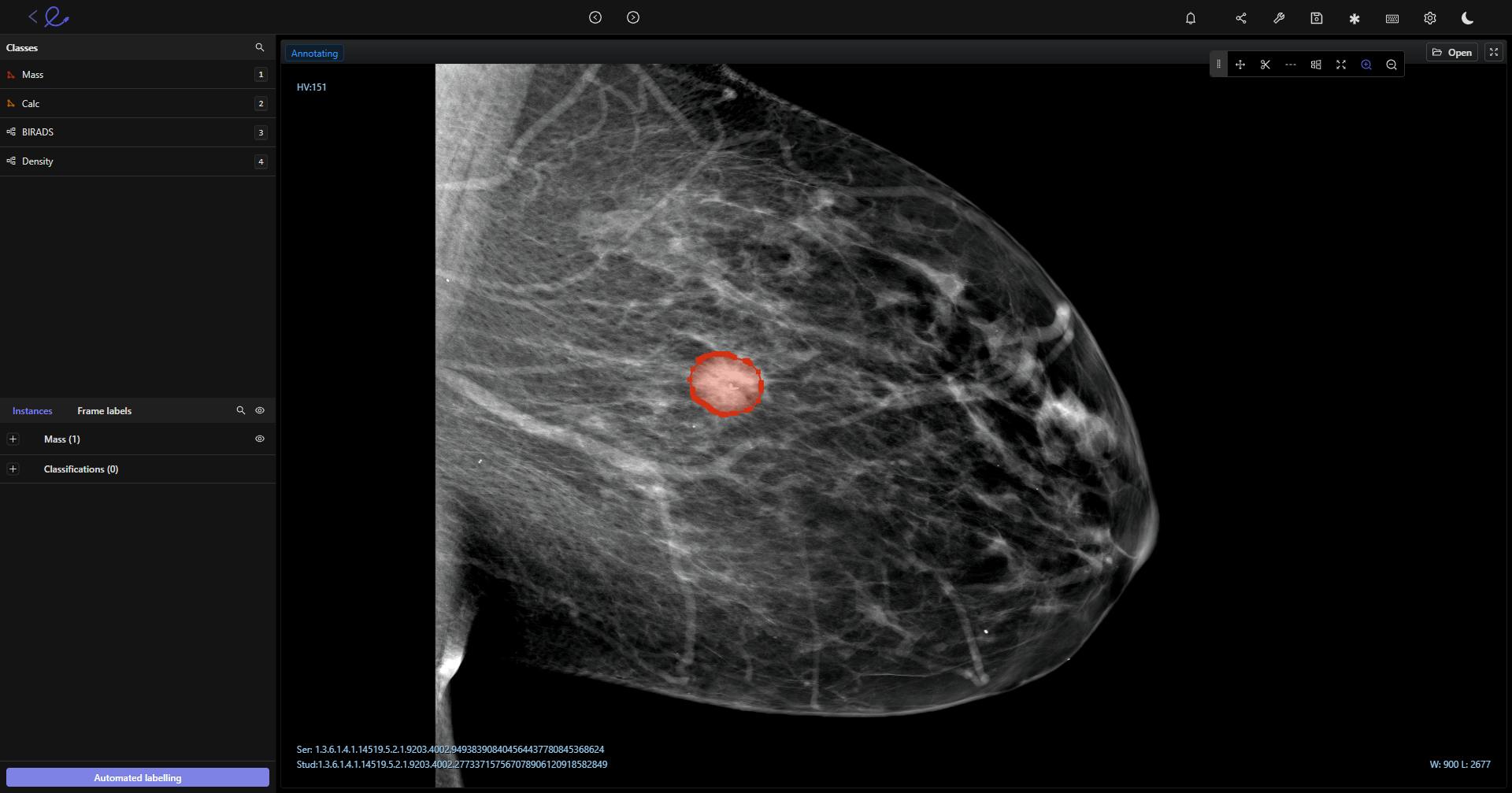
An example of DICOM annotation in Encord
Alongside these complications, project leaders have to weigh the necessity of gaining regulatory approval for working models, clear audit trails, and enhanced data security. Remember, the ultimate outcome of any medical machine learning model could directly impact patient healthcare treatment outcomes. Accuracy and keeping bias as low as possible are essential.
For example, a slight inaccuracy when analyzing music preference data isn’t going to hurt anyone. Whereas, with medical imaging datasets, the accuracy and results can have serious, life-changing outcomes for patients worldwide. Hence the need to test as much data as possible. Not only is this important to ensure a robust model for primary use cases; but you also need to assess datasets and models against a wider range of edge and corner cases.
Machine learning medical imaging dataset experiments have tried and tested workflows that improve efficiency. Before starting ML-based experiments, you need to ensure you’ve got the right components to start running experiments.
Components of a machine learning experiment workflow need to include:
Once these components are ready, you can start running machine-learning experiments on medical imaging datasets. An ideal workflow should involve the following:
With machine learning experiments, or any computer vision or AI-based experiments, the more data you have the better. Especially when it comes to medical imaging ML model experiments for most use cases.
However, it’s important to remember that quality and diversity are as important as the volume of data. Medical imaging data should include the most relevant clinical practice data possible for the experiments. Such as having enough images with positive and negative cases, different ethnic groups, and either including or excluding the relevant edge cases; e.g. patients who have or haven’t received treatment.
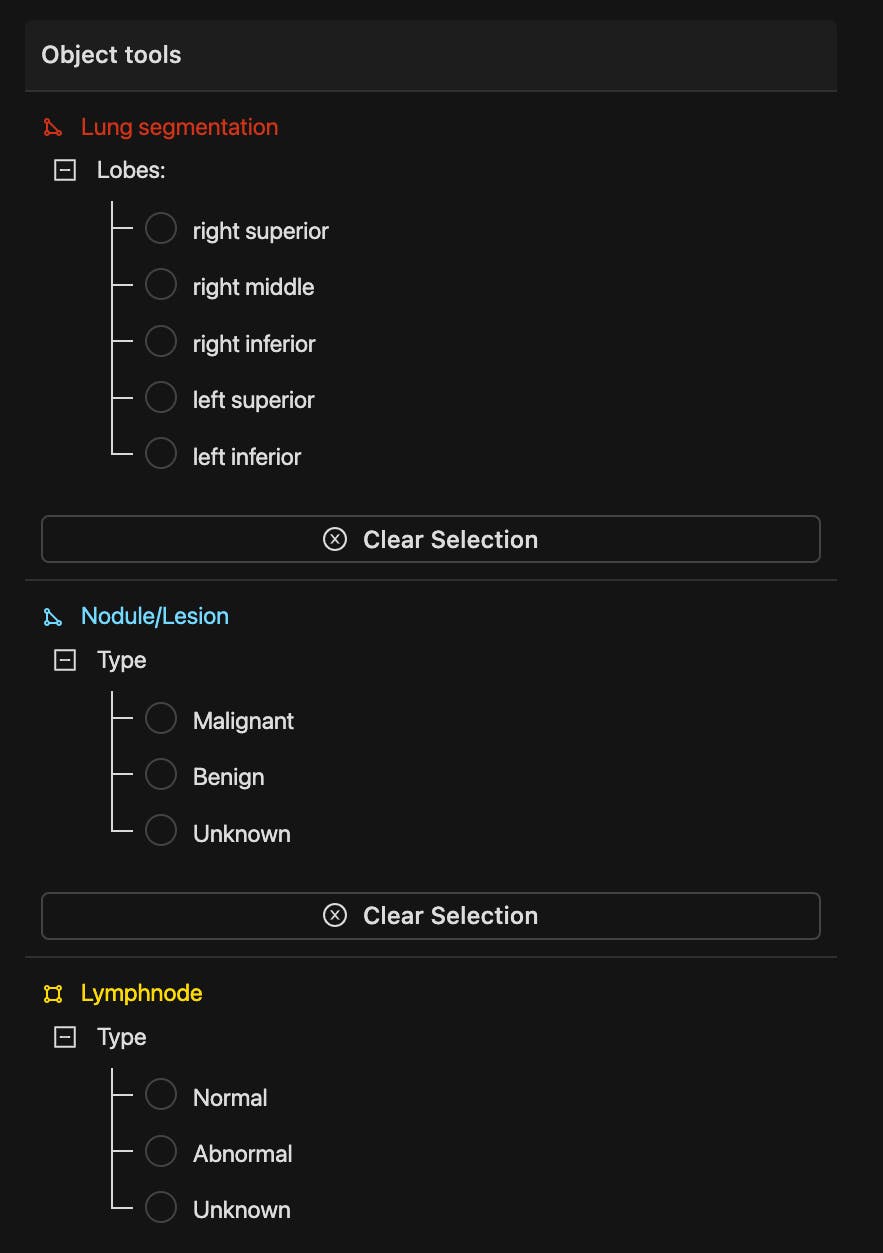
Example of a DICOM image ontology in Encord
Getting a high volume of data is crucial. But the quality and diversity of the datasets you’ve got available matter too. As does the quality and accuracy of the annotations and labels applied and reviewed by skilled radiologists and clinicians.
During machine learning experiments, most go wrong or fail in some way. That’s not unusual. As most data scientists and clinical ops managers know, this is normal. You might have 100 experiments running and only 10 to 15 produce outcomes close to what you need. A failure isn’t a setback.
In fact, following the scientific methodology, failures simply get you closer to successful outcomes that validate a hypothesis. Even if a hypothesis is invalidated, that’s a positive too, as it will help you refocus efforts on the right perimeters and valuators to test a new theory. Or in some cases, a negative outcome could be the goal behind an ML-based experiment.
So, it’s useful to never see failure as a negative but to learn from the experiments that fail and move forward with the learnings from those that have achieved the desired outcomes.
Only this way can you successfully put a machine learning model into production.
With the right tools, processes, and systems, project and clinical ops managers can create efficient medical imaging machine learning project workflows.
Open-source tools can be a great starting point but can make it harder to develop the scope of your projects. For example, open-source tools can reduce efficiency, make scaling difficult, weaken data security, and monitoring or audit annotators’ work is almost impossible.
Instead, medical image dataset, annotation, and machine-learning teams benefit from using proprietary automated image annotation tools to improve experiment efficiency.
Encord has developed our medical imaging dataset annotation software in close collaboration with medical professionals and healthcare data scientists, giving you a powerful automated image annotation suite, fully auditable data, and powerful labeling protocols.
Ready to automate and improve the quality of your medical data annotations?
Sign-up for an Encord Free Trial: The Active Learning Platform for Computer Vision, used by the world’s leading computer vision teams.
AI-assisted labeling, model training & diagnostics, find & fix dataset errors and biases, all in one collaborative active learning platform, to get to production AI faster. Try Encord for Free Today.
Want to stay updated?
Follow us on Twitter and LinkedIn for more content on computer vision, training data, and active learning.
Join the Encord Developers community to discuss the latest in computer vision, machine learning, and data-centric AI
Join the communitySoftware To Help You Turn Your Data Into AI
Forget fragmented workflows, annotation tools, and Notebooks for building AI applications. Encord Data Engine accelerates every step of taking your model into production.